Water and potassium reaction12/27/2023 
It also vigorously reacts with water and catches fire. Same nature is also shown by another alkali metal that is sodium (Na). This reaction is so fast and exothermic that it can even take place in cold water as well. Conclusion from the above data is that potassium reacts with water to produce lilac-colored flame. Sometimes we can also see explosions at the end of the reaction. The flame produced is a lilac-colored flame. This reaction is so vigorous and exothermic that hydrogen gas liberated during the reaction catches fire spontaneously. Strongly alkaline potassium hydroxide is formed in solution. Hydrogen is produced, and with potassium vapor, burns with a pink or lilac flame. 2 K(s) + 2 H 2 O(l) 2 KOH(aq) + H 2 (g) A reaction of potassium metal with water. The other example of a precipitation reaction is the reaction between calcium chloride and potassium hydroxide, resulting in the formation of an insoluble salt Called calcium hydroxide. Potassium metal can react violently with water producing KOH and hydrogen gas. Precipitation reactions help in determining the presence of different ions present in a particular solution. Here we can see that when potassium reacts with water, it produces potassium hydroxide along with hydrogen gas. This solid silver chloride is insoluble in water. As potassium is highly reactive, so when it reacts with water following reaction takes place:. Sodium moves because of the hydrogen coming off of it and has a low melting point while potassium’s reaction is faster and enough heat is produced. 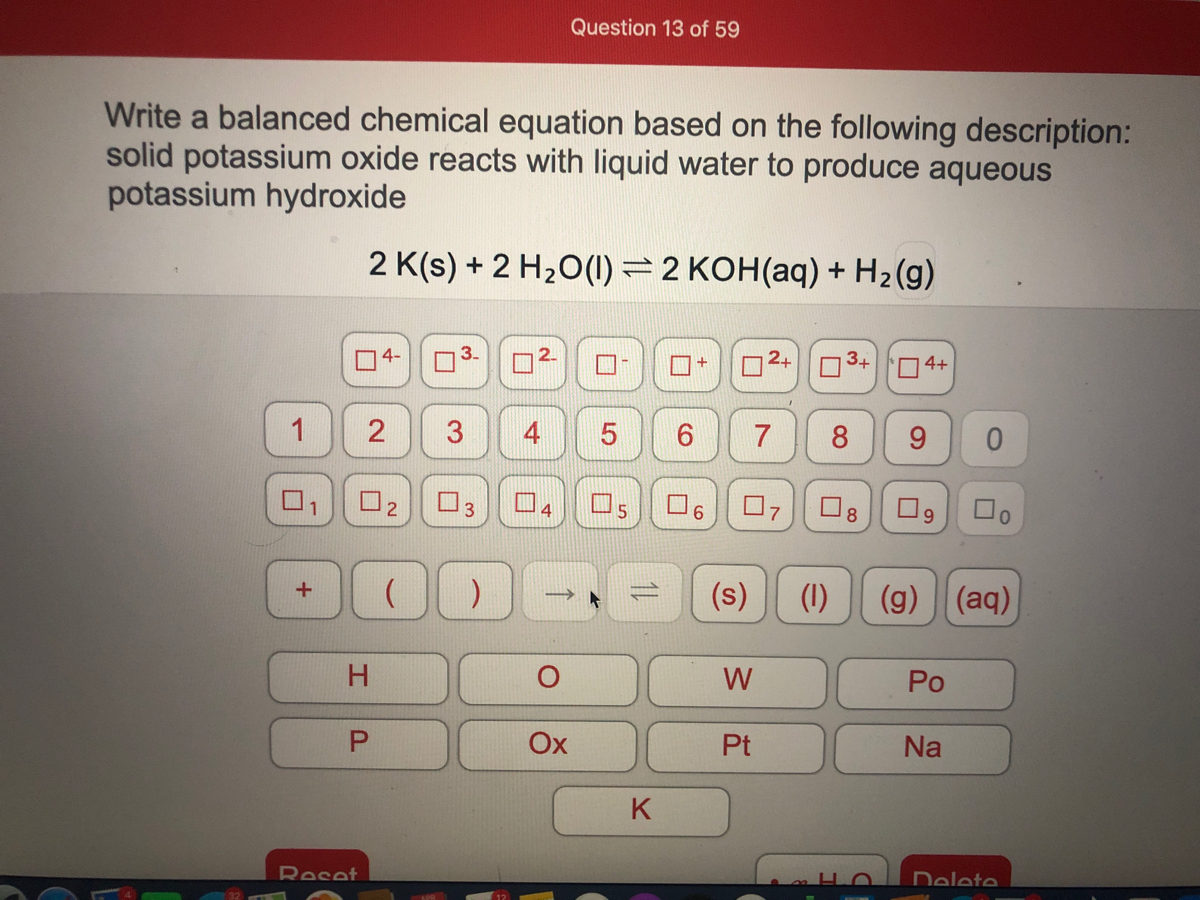
Sodium and potassium react somewhat similar when reacting with water however they are slightly different. It can combine with anions to form ionic salts and potassium generally occurs only in form ionic compounds in nature. How would you compare that relative reactivity of Na and k. 
In the periodic table, potassium belongs to the alkali metals group, all of them have a single valence electron in the outermost shell which can be easily removed to create an ion with a positive charge (a cation). It is a silvery-white metal which is so soft enough to be cut with a knife just by a little force. Potassium: It is a chemical element denoted by the symbol ‘K’ and has atomic number 19. Let us first understand about potassium metal and its nature followed by the type of fame get produced when it reacts with water as follows:. So here we have to tell what type of flame gets produced when potassium reacts with water. The chemical equation for the reaction of potassium with water is 2K(s) + 2H2O(l) 2KOH(aq) + H2(g). This metal moves around quickly on the water and spreads the fire everywhere. When it is added to water, the potassium metal floats and melts as well as sets on fire. Hint: As we know that potassium is an alkali metal and very reactive in nature.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
